TBSend card
A breakthrough electricity-free device for safe sputum transport, long-term storage, and rapid DNA release for tuberculosis molecular testing. Seamlessly compatible with Truenat, GeneXpert, and other CB-NAAT/NAAT platforms, it delivers high-quality DNA through a simple manual bead-based extraction—making the process entirely power-independent and field-ready.
The device has cleared biosafety validation at ICMR–NIRT, Chennai, sponsored by the India Health Fund (Tata Trusts), and is entering Phase II efficacy evaluation by ICMR.
Recognized by The Union’s Stop TB–Build initiative among India’s Top 5 TB innovations, the TB Send Card exemplifies India’s leadership in accessible, field-adaptable molecular diagnostics.

Figure : The TB Send Card device and its key components — including reagent vials, storage casket, and biosafe card unit — enable safe sputum transport, ambient storage, and rapid DNA release for molecular TB testing without electricity.

Figure : The image shows the TB Send Card storage casket (black cylindrical unit) placed on custom-designed 3D-printed support holders used for safe positioning during sample application, drying, and storage.
TB Send Card Device Demo
Fluorescent Probing based MTB Drug Resistance using Contact Quenchers
- Detection of all common Mycobacterium tuberculosis MDR mutations & major XDR mutations using contact quenching technology and wild allele blockers.
- Highly economical.
- Wide coverage of mutations.
- Genetically engineered controls for all mutations constructed specially for this kit.
- Integrated PCR Inhibition & generic tuberculosis detection module.
- A Wobble Base Innovation.
- Research funded by Biotechnology Industry Research Assistance Council (BIRAC), Govt. of India under the Small Business Innovation Research Initiative (SBIRI)
Hybrid Spin Columns
- A spin column containing multiple layers of membranes of different compositions for an ultra efficient purification of nucleic acids.
- Patented (Patent Granted)
- Highly economical.
- An excellent substitute for imported spin columns.
- Comes along with synthetic flocculating reagent that is an economical substitute for transfer RNA used for efficient precipitation of nucleic acids.


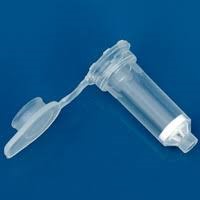
Figure : Images of hybrid Spin columns (Patent granted) along with collection tubes. The blue coloured column is specialized for RNA extraction.
Chromosomal DNA- shearing solution and tRNA analog in Human Blood RNA Extraction
- An efficient and very cost effective set of reagents for extraction of total RNA from human blood cells.
- Use of an innovative shearing solution that effectively shears human chromosomal DNA that aids in its efficient removal from the solution.
- Chromosomal RNA is removed through physical shearing rather than by an enzymatic process as seen in some popular commercial kits.
- Use of a specialized column, in addition to hybrid spin columns, that remove debris prior to purification of RNA.

Figure : Chromosomal DNA shearing solution presented in a convenient dropper bottle that releases 12 uL of suspension per drop. The suspension not only shears chromosomal DNA to allow its effective removal but also stabilizes the RNA and preserve its integrity for downstream applications.
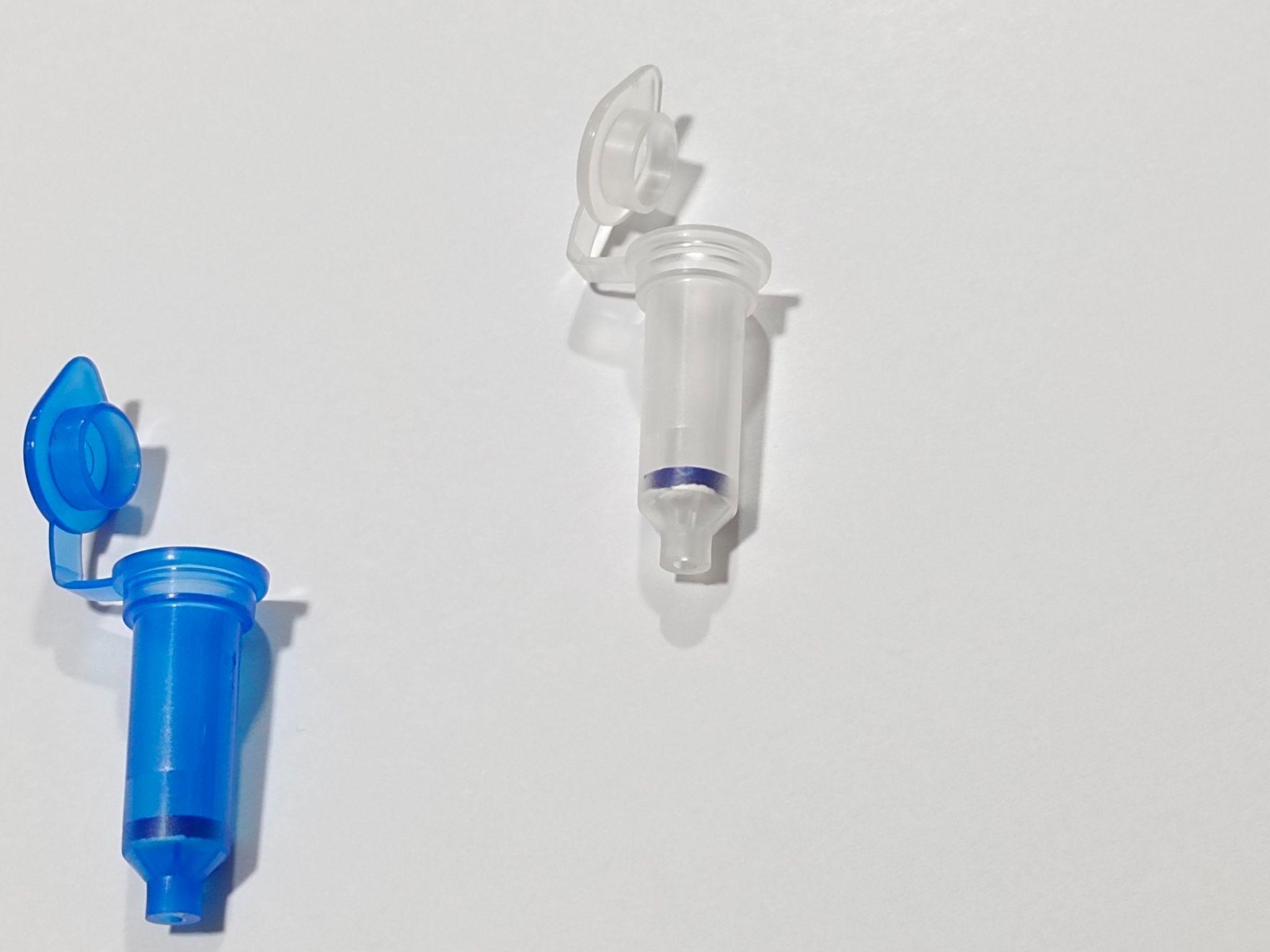
Figure : A pair of spin columns. the blue colored column designed to remove chromosomal debris while the white colored hybrid spin column (Patent granted) designed to purify the RNA.
Repurposing a glucometer for viral load detection
- A novel method of detecting genome copy numbers using glucometer as the detection platform.
- detection primers are tagged with Invertase enzyme.
- Isothermal exponential amplification allows accumulation of enzyme tagged primer ends in the solution.
- The resultant solution is incubated in a pure solution of sucrose.
- Accumulated invertase enzymes convert sucrose to glucose that is detected by a glucometer.
- The conversion of sucrose to glucose linearly correlates with accumulated enzymes tagged at each end of an amplification product and is demonstrated to be a surrogate marker for copy number of the template amplified.
- The technology is at its validation stage and has potential to detect copy number of any DNA or cDNA template amenable to isothermal or end point PCR based amplification.





Awards and Grants
It’s always a team efort towards improving the health of our nation. We are dedicated to bring innovative solutions to people in every part of the world.

